 
In essence, #"Ar"# is just a representation for #1s^(2)2s^2 2p^6 3s^2 3p^6# without us having to write it all out.Īll in all, the three given answers are correct ways of figuring out the ground-state electron configuration of Krypton. Cesium has the smallest ionization energy of all elements (376 kJ/mol), and chlorine has the most negative electron affinity 1-349 kJ/mol2. *Notice how the configuration #1s^(2)2s^2 2p^6 3s^2 3p^6# is the electron configuration for #"Ar"# so by starting with #"Ar"# we indicate that it is the configuration of everything that came before. The end result would then be: * # 4s^2 3d^10 4p^6# From there we write out the remaining configuration like we did in the first method. But we could also write out the configuration beginning using #"Ar"# since it is the closest noble gas with the lower energy. This method becomes really useful for elements that have a lot of electrons like Krypton because it becomes a hassle to write out such a long electron configuration.Ĭoincidently, Krypton itself is a noble gas so we could write the electron configuration as #. In essence, the shorthand notation tells us the configuration by using a noble gas element as our starting point instead of starting all the way at the #1s# orbital. The key to using this method is to identify the noble gas closest to the desired element that is at a lower energy (Has a lower atomic number if I'm loosely speaking). Using this as a guide and going through the periodic table we find the electron configuration to be: The element cesium (or caesium, atomic number 55) has the electron configuration 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s1 or abbreviated as Xe 6s1 Wiki User 13y ago This answer. We know that the #s,p,d# orbital can hold a max of #2,6,10# electrons respectively. One way is to write out the entire electron configuration by going through each orbital or we can use a shorthand notation using the noble gases as a starting point. Third: Write out the electron configurationįor Krypton and most of the elements there are more the just one way (usually two) to write the electron configuration. This electron configuration shows that the indium ion (In 3+) has four shells and the last shell has eighteen electrons and it achieves stable electron configuration. The electron configuration for the first 10 elements. The electron configuration of indium ion (In 3+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10. (Refer to the following pictures as notes) Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. Know how many electrons each orbital can hold and their order. The item number off television is 52.First: Determine the number of electrons. Cesium, chemical element of Group 1 (also called Group Ia) of the periodic table, the alkali metal group, and the first element to be discovered spectroscopically (1860), by German scientists Robert Bunsen and Gustav Kirchhoff, who named it for the unique blue lines of its spectrum (from the Latin caesius, ‘sky-blue’). It is having eight electrons and neutrons state and the electronic configuration is one is to do this, too, to be for and for telegram. 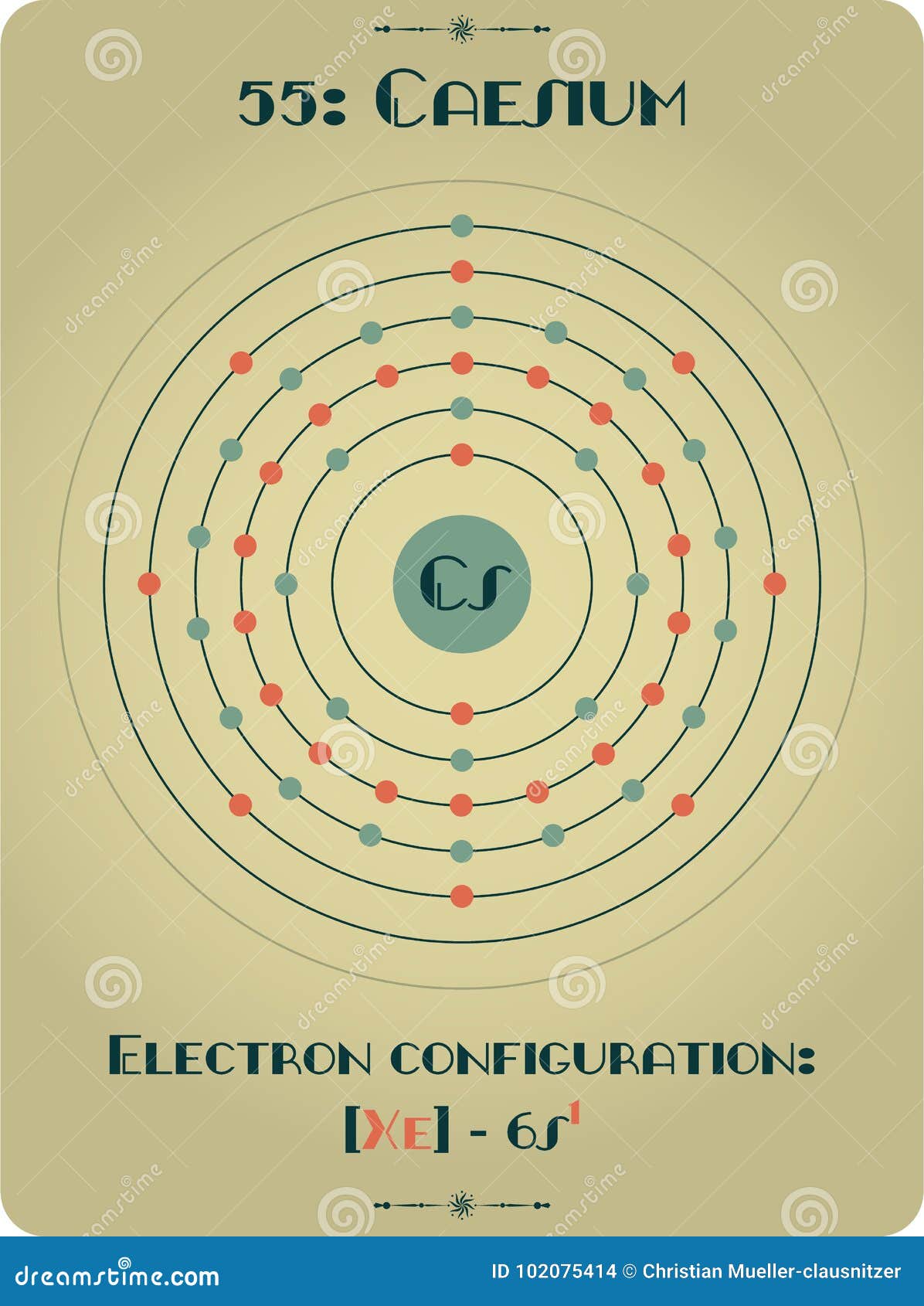
Where is the outermost shell off? Caesium is six. The hard shell is the outermost shell for sodium. 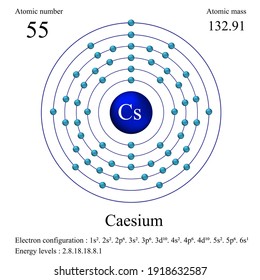
However, we see that that the outermost shell off sodium is three. The outermost shell is having one electrons for both. The complete electronic configuration of Cs is 1s22s22p63d63d104s24p64d105s25p66s1 and with noble gas coore is Xe6s1.They're having only one electron in the outermost shell. Now here we can see that put sodium and see them. So this is the electron configuration for caesium. And that number for Susan is 55 latest right down the electron configuration for caesium. And finally, electron will goto the most 73 years one. Let's strike down the electron configuration for sodium, which is one is to too, and two, two p six. Therefore, sodium is having 11 electrons in its natural state. We need to find the similarities and difference in the electron configuration off the parents that given are strongly sodium and Susie, um, sodium. Name: Cesium Symbol: Cs Atomic Number: 55 Atomic Mass: 132.90546 amu Melting Point: 28.5 ☌ (301.65 K, 83.3 ☏) Boiling Point: 678.4 ☌ (951.55005 K, 1253.12 ☏) Number of Protons/Electrons: 55 Number of Neutrons: 78 Classification: Alkali Metal Crystal Structure: Cubic Density 293 K: 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
